Physiology Assignment on Urinalysis & Digestive Enzymes
Question
Task: You must provide short answers containing at least six sentences to the following questions. You must use scientific textbooks and scientific research papers to reference your report.
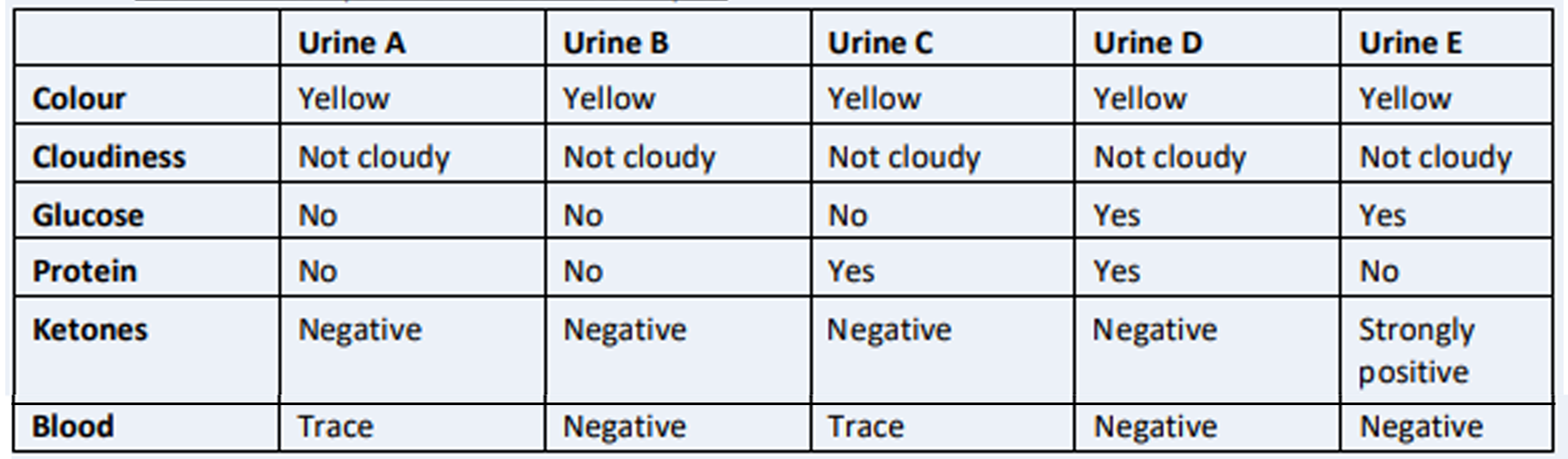
1. Urinalysis is a technique involving physical, chemical and microscopic analyses of a sample of urine. Table 1 describes the physical and chemical properties of five samples of patients’ urine.
a) Which sample represents a healthy patient? Explain and reference your answer
b) Which of these patients is diabetic? Explain and reference your answer
Table 1: Descriptions of five urine samples
2. The bicinchoninic acid (BCA) protein assay enables the quantification of protein by measuring the absorbance of a BCA : protein complex at 562nm. Below is a table containing absorbance readings at 562 nm for standard protein samples of known protein concentrations.
Table 2: Absorbance readings at 562 nm for six standard and two test samples
a) Calculate the mean absorbance reading for each sample (standards 1 – 6 and test samples 1 and 2). Show all your working and present your data in table format.
b) Calculate the normalized absorbance reading for each standard sample by subtracting the mean value for sample 6 from each sample’s mean value. Show all your working and present your data in table format.
c) Why is it necessary to subtract the mean value for standard 6 from each sample’s mean absorbance reading?
d) What happens to the absorbance values at 562 nm as the protein concentration of the standard samples increases?
e) Why are you measuring the absorbance of these samples at 562 nm and not 280 nm? Reference your answer
3. Plot a graph where the x-axis represents Protein Concentration (?g / ml) and the y-axis represents Normalized Absorbance values at 562nm. Work out the equation for the line of best fit.
4. Using the equation of the line of best fit, calculate the concentrations of proteins in test samples 1 and 2. Show all your working.
5. During normal digestion what is the location and function of the enzyme salivary amylase? Reference your answer
6. What pH and temperature does salivary amylase function at? Reference your answer 7. Experimentally the presence of starch is determined using the Lugol’s test and the appearance of maltose is determined by the Benedict’s test. Four solutions were tested for the presence of starch and maltose using these tests. The results are listed in table 3.
For the Benedict’s test a blue colour indicated the absence of maltose
a green colour indicates the presence of a tiny amount of maltose
a yellow colour indicates the presence of some maltose
a red colour indicates the presence of a lot of maltose
For the Lugol’s test a purple – black colour indicates the presence of a lot of starch
a purple – blue colour indicates the presence of a small amount of starch
a yellow – orange indicates the absence of starch
Using this information deduce which tube(s) contained the most starch following incubation?
8. Which tube(s) contained the most maltose?
9. What conclusions do you draw from these results?
10. What was the purpose of carrying out the Lugol’s and Benedict’s tests in the presence of water?
Table 3: Carbohydrate digestion of four experimental samples
Answer
Answer 1.
A)The main function of the kidney identified in this context of physiology assignment is to filter the waste material out of the body in the form of urine whose characteristics determine the underlying health issue. Research on physiology assignment suggests that colour of urine is dependent on the breakdown of the product of RBC that further converts into water-soluble forms and eliminates into the bile; thus, it imparts yellow colour (Kalakondaand John 2019). In the case scenario of physiology assignment, the five samples of urine were reported with yellow colour and non-cloudiness characteristic which shows normal features; hence it was further analysed for the presence of protein, ketones, glucose and blood. In a normal healthy human, during the filtration process of the glomerulus, protein is unable to move out of the glomerulus capillaries and hence it is absent or present in small traces in urine (Callensand Bartges 2015). The sample C and D show presence of protein therefore this sample does not belong to a healthy person.
Fat metabolism gives ketones where the body uses fats as the source of energy. A healthy human uses glucose as the source of energy therefore it is absent in urine. Sample E shows a high concentration of ketones hence does not represent a healthy patient. The research by Bondonno et al. (2015) considered in this section of physiology assignment has indicated that blood and glucose are absent in the urine of healthy individuals due to the fact of metabolism of glucose that gets utilised by the body and absorbed by kidney and traces of blood show damage to kidney or infection. Sample A shows the presence of blood in the urine and sample B is negative to both glucose and blood as well as has a yellow colour, not cloudy and absence of ketones. Therefore, Urine sample B represents a healthy patient.
B) The urine test for the diabetic patient is confirmed mainly from the presence of ketones and glucose (Peters et al. 2016). In a diabetic patient, the glucose is not able to get transported to the body due to the absence of insulin that leads to high blood sugar level and causesits spilling over into the urine (Peters et al. 2016). Moreover, in the diabetic patient due to resistant to insulin or low secretion, the high amount of glucose in the bloodstream and low concentration in body cells cause to use fats as the source of energy. Research on physiology assignment has shown that the storage of fats and its metabolism causes the production of ketones as the by-product and ultimately its concentration in urine appears to be high in diabetic patients (Yousri et al. 2015). Therefore, Urine sample E represents a high concentration of ketones and the presence of glucose suggesting it to be of a diabetic patient.
Answer 2
Table 1: Mean Absorbance readings at 562 nm for six standards and two test samples
|
Sample tube |
Absorbance reading 1 |
Absorbance reading 1 |
Absorbance reading 1 |
Protein concentration (?g / ml) |
Mean absorbance |
|
Standard 1 |
1.242 |
1.356 |
1.081 |
1000 |
|
|
Standard 2 |
0.847 |
0.909 |
0.923 |
500 |
|
|
Standard 3 |
0.494 |
0.493 |
0.555 |
250 |
|
|
Standard 4 |
0.397 |
0.373 |
0.401 |
125 |
|
|
Standard 5 |
0.267 |
0.247 |
0.262 |
25 |
|
|
Standard 6 |
0.237 |
0.224 |
0.229 |
0 |
|
|
Test sample 1 |
1.242 |
1.356 |
1.081 |
Unknown |
|
|
Test sample 2 |
0.397 |
0.373 |
0.401 |
Unknown |
|
A) The mean absorbance reading standard 1:
The mean absorbance reading standard 2:
The mean absorbance reading standard 4
The mean absorbance reading standard 4
The mean absorbance reading standard 5
The mean absorbance reading standard 6
The mean absorbance reading test sample 1
The mean absorbance reading test sample 2
B) Normalized absorbance reading of standard sample 1: 1.22 – 0.23 = 0.99
Normalized absorbance reading of standard sample 2: 0.893-0.23 = 0.663
Normalized absorbance reading of standard sample 3: 0.514- 0.23 = 0.284
Normalized absorbance reading of standard sample 4: 0.390 – 0.23 = 0.16
Normalized absorbance reading of standard sample 5: 0.258- 0.23 = 0.028
Normalized absorbance reading of standard sample 6: 0.23 - -0.23 = 0
Normalized absorbance reading of test sample 1: 0.893-0.23 = 0.663
Normalized absorbance reading of test sample 2: 0.390 – 0.23 = 0.16
Table 2: Normalised Absorbance readings at 562 nm for six standard and two test samples
|
Sample tube |
Absorbance reading 1 |
Absorbance reading 1 |
Absorbance reading 1 |
Protein concentration (?g / ml) |
Normalised absorbance |
|
Standard 1 |
1.242 |
1.356 |
1.081 |
1000 |
0.99 |
|
Standard 2 |
0.847 |
0.909 |
0.923 |
500 |
0.663 |
|
Standard 3 |
0.494 |
0.493 |
0.555 |
250 |
0.284 |
|
Standard 4 |
0.397 |
0.373 |
0.401 |
125 |
0.16 |
|
Standard 5 |
0.267 |
0.247 |
0.262 |
25 |
0.028 |
|
Standard 6 |
0.237 |
0.224 |
0.229 |
0 |
0 |
|
Test sample 1 |
1.242 |
1.356 |
1.081 |
Unknown |
0.663 |
|
Test sample 2 |
0.397 |
0.373 |
0.401 |
Unknown |
0.16 |
C) Sample 6 shows the absence of any protein and function as a blank sample in the bicinchoninic acid (BCA) protein assay. The mean value of sample 6 shows the absorbance value of component other than protein hence its subtraction from the rest of the sample will give accurate protein absorbance value by eliminating other factors. Thus, it controls variation in the result and lowers the measurement error.
D) The absorbance value of the standard samples increases with an increase in protein concentration at 562 nm that satisfies the lambert beer law A=?bc, where b = path length, c = concentration of the absorbing sample, and ? = constant of proportionality.
E) The sample analyzed is a bicinchoninic acid assay that depends on the conversion of the Copper ion to Cu1+ under the alkaline solution as depicted in the below figure provided within this physiology assignment.
Figure 1. Schematic representation of BCA assay.
Source: (Johnson 2016)
The bicinchoninic acid (BCA) assay is greatly influenced by the presence of the amino acid residues and the is a definite reagent for the Cu1+. In this physiology assignment, the research by Johnson(2016) has indicated that the concentration of Cu1+ and protein concentration are a directly proportional and dependent factor which is determined spectrophotometrically and imparting color change from blue to purple. Its Copper ion is known to absorb the light at the wavelength of 562 nm therefore absorbance of standard samples is taken at 562 nm and not 280 nm.
Answer 3.
The equation for the line of best fit is y = 0.001x + 0.0318
X is the unknown concentration and the Y value is the absorbance
Answer 4.
Using the y = 0.001x + 0.0318, the concentration of protein for the test sample 1 is
0.001x= y - 0.0318
0.001x= 0.663 - 0.0318
0.001x = 0.6312
X =
Concentration of protein for the test sample 1 =
The concentration of test sample 2
0.001x= y - 0.0318
0.001x = 0.16 – 0.0318
0.001x = 0.1282
X =
The concentration of test sample 2 =
Table 3: Protein concentration of two test samples
Sample tube Absorbance reading 1 Absorbance reading 1 Absorbance reading 1 Protein concentration (?g / ml)
Standard 1 1.242 1.356 1.081 1000
Standard 2 0.847 0.909 0.923 500
Standard 3 0.494 0.493 0.555 250
Standard 4 0.397 0.373 0.401 125
Standard 5 0.267 0.247 0.262 25
Standard 6 0.237 0.224 0.229 0
Test sample 1 1.242 1.356 1.081 631.2
Test sample 2 0.397 0.373 0.401 128.2
Answer 5.
Salivary Amylase is the active digestive enzyme present in the mouth, secreted from parotid glands which are located below and in the front of each ear canal. The primary function of the salivary amylase presented in the physiology assignment is to act on starch and break down the carbohydrates into smaller molecules which can be easily absorbed (Boehlke,Zierauand Hannig 2015). It eases the digestion process by cleaving the starch into dextrin and further into maltooligosaccharides and isomaltooligosaccharides. Hence, it can be said that it converts starch to maltose and complete the digestion of the carbohydrates releasing glucose that is also absorbed into the blood. The studies have suggested that hydrolysis of starch by the action of the enzyme amylase takes place in seconds in mouth and it causes the transformation of the gelatinous nature of the starch into a semi-liquid state (Dawes et al. 2015). Such transformation influences the digestion of starch and its consumption hence such evidence suggests a pre-absorptive role of the salivary amylase.
Answer 6.
The action and function of salivary amylase are greatly influenced by the temperature and pH and have specific pH and temperature for optimum activity. The research carried on this physiology assignment has shown that due to the proteinaceous nature of the salivary amylase, lower temperature deactivates the enzyme and higher temperature cause denaturation of the protein (Jain,Jainand Jain 2020). It suggests that the digestion of the starch will take a longer time at the lower and higher temperature. Therefore, the optimum temperature for the function of amylase ranges from 32 °C to 37 °C.
Similarly, the level of pH also affects the time of action of salivary amylase. The optimum pH for the action of the salivary enzyme is 6 to 7 as research has shown that pH below 6 and above 7 lowers the rate of reaction of the enzymes and eventually causes the protein to be denatured (Owen 2019). In this physiology assignment, research has also suggested that at a pH of 6.8, the salivary amylase enzyme is most active due to the neutral environment that maintains the structure of the amylase ((Jain,Jainand Jain 2020).
Answer 7.
Research on physiology assignment has noted that starch is a complex carbohydrate that breaks into maltose in the simplest sugar which is tested throughLugol’s test (Low et al. 2017). The presence of starch imparts variation in the color sample that shows the presence and absence of the sugar. In tube 1 which contains starch and water gives a purple-black color on Lugol’s test suggesting the presence of a high amount of starch. Since water does not cause cleavage of the starch molecules its concentration is high. However, when compared with other tubes samples like starch and saliva of tube 2 it can be interpreted that saliva having digestion activity breaks the starch molecules present in the tube into simple sugar and causes its absence which is evident from yellow-orange color (Boehlke,Zierauand Hannig 2015). Similarly, other tubes number 3 and 4 containing Hydrochloric acid and saliva; and boiled salivabreaks small amounts of starch due to loss of action of saliva in presence of Hydrochloric acid and denatured nature of boiled saliva respectively. Therefore, it can be said herein physiology assignment that tube 1 contains lots of starch.
Answer 8.
The benedict test carried out to test maltose by giving a red color to the solution. The tube 1 containing starch and water, does not cause breakage of starch into maltose thus imparting blue color that shows an absence of maltose. The tube 2 containing starch and sugar gives a red color that depicts the high amount of maltose is articulated from the function of saliva in digestion of the starch into maltose. In tube 3 containing starch, saliva and HCL; though saliva cause breakage of starch into maltose (Freitas et al. 2018) it does not show its function in presence of HCL causing less amount of maltose evident from orange color. Similarly, tube 4, containing starch and boiled saliva shows yellow color suggesting a small amount of maltose associated with loss of function of saliva due to the high temperature that causes its denaturation. Therefore, it can be said herein physiology assignment that tube 1 contained most maltose.
Answer 9.
From the gathered result it is concluded that starch is the complex molecules and breaks into maltose on hydrolysis in the presence of saliva. The reaction of the test inferred that saliva has the enzyme that causes the breaking of the starch molecules and its activity is highly affected by hydrochloric acid as it tends to increase its pH values of the solution (Boehlke,Zierauand Hannig 2015). Moreover, it can also be concluded from the above discussion on physiology assignment that temperature has a great influence on the action of saliva as it causes its denaturation and does not cause the conversion of starch into maltose (Dawes et al. 2015).
Answer 10.
The purpose of carrying out Lugol’s and Benedict’s tests within this physiology assignment in the presence of water is to confirm the digestive role of saliva upon starch by comparing the result of the presence of starch in water. The information provided in the physiology assignmentalso shows that result is not affected by any other protein molecules, thus act as a positive control in the test.
?
Reference
Boehlke, C., Zierau, O. and Hannig, C., 2015. Salivary amylase–The enzyme of unspecialized euryphagous animals.
Physiology assignmentArchives of oral biology, 60(8), pp.1162-1176.
Bondonno, C.P., Liu, A.H., Croft, K.D., Ward, N.C., Shinde, S., Moodley, Y., Lundberg, J.O., Puddey, I.B., Woodman, R.J. and Hodgson, J.M., 2015. Absence of an effect of high nitrate intake from beetroot juice on blood pressure in treated hypertensive individuals: a randomized controlled trial. The American journal of clinical nutrition, 102(2), pp.368-375.
Callens, A.J. and Bartges, J.W., 2015. Urinalysis. Veterinary Clinics: Small Animal Practice, 45(4), pp.621-637.
Dawes, C., Pedersen, A.M.L., Villa, A., Ekström, J., Proctor, G.B., Vissink, A., Aframian, D., McGowan, R., Aliko, A., Narayana, N. and Sia, Y.W., 2015. The functions of human saliva: A review sponsored by the World Workshop on Oral Medicine VI. Archives of oral biology, 60(6), pp.863-874.
Freitas, D., Le Feunteun, S., Panouillé, M. and Souchon, I., 2018. The important role of salivary ?-amylase in the gastric digestion of wheat bread starch. Food & function, 9(1), pp.200-208.
Jain, A., Jain, R. and Jain, S., 2020.Study of Enzymatic Hydrolysis of Starch.In Basic Techniques in Biochemistry, Microbiology and Molecular Biology (pp. 221-222).Physiology assignmentHumana, New York, NY.
Jain, A., Jain, R. and Jain, S., 2020. Study the Effect of Temperature on Salivary Amylase Activity. In Basic Techniques in Biochemistry, Microbiology and Molecular Biology (pp. 227-229).Humana, New York, NY.
Johnson, M. (2016).Protein Quantitation. Materials And Methods, 2. doi: 10.13070/mm.en.2.115
Kalakonda, A. and John, S., 2019. Physiology, Bilirubin.In StatPearls [Internet].StatPearls Publishing.
Low, J.Y., Lacy, K.E., McBride, R.L. and Keast, R.S., 2017.Evidence supporting oral sensitivity to complex carbohydrates independent of sweet taste sensitivity in humans. PloS one, 12(12). Owen, M., 2019.Amylase activity–A microscale approach in biology. African Journal of Chemical Education, 9(3), pp.64-74.
Peters, A.L., Henry, R.R., Thakkar, P., Tong, C. and Alba, M., 2016.Diabetic ketoacidosis with canagliflozin, a sodium–glucose cotransporter 2 inhibitor, in patients with type 1 diabetes. Diabetes Care, 39(4), pp.532-538.
Yousri, N.A., Mook-Kanamori, D.O., Selim, M.M.E.D., Takiddin, A.H., Al-Homsi, H., Al-Mahmoud, K.A., Karoly, E.D., Krumsiek, J., Do, K.T., Neumaier, U. and Mook-Kanamori, M.J., 2015. Physiology assignmentA systems view of type 2 diabetes-associated metabolic perturbations in saliva, blood and urine at different timescales of glycaemic control. Diabetologia, 58(8), pp.1855-1867.